 Anette Duensing, MD is a research pathologist who is a highly valued member of the Science Committee of GIST Support International. A native of Germany, Dr. Duensing did a post-doc at the Dana-Farber lab of Jonathan Fletcher, MD before joining the faculty at the University of Pittsburgh.
Anette Duensing, MD is a research pathologist who is a highly valued member of the Science Committee of GIST Support International. A native of Germany, Dr. Duensing did a post-doc at the Dana-Farber lab of Jonathan Fletcher, MD before joining the faculty at the University of Pittsburgh.
New Findings on GIST Quiescence
Dr. Anette Duensing and her colleagues have published a new paper describing how some GIST cells become dormant or quiescent in response to imatinib (Gleevec). Just printed in Cancer Research on November 1, the paper by Liu et al is entitled “Imatinib Mesylate Induces Quiescence in GIST Cells Through the CDH1-SKP2-p27Kip1 Signaling Axis.†Dr. Duensing continues to unravel the underlying mechanisms that determine whether GIST cells die from imatinib. In this paper she details that some cells evade death by becoming quiescent. Earlier Dr. Duensing had published about the exact mechanism through which imatinib can cause GIST cells to die via programmed cell death (apoptosis) triggered by histone H2AX [Liu et al, Cancer Research 2007, 67:2685-2692]. She hopes that finding will lead to a way to boost imatinib effectiveness by increasing H2AX quantities, thereby promoting cell death. However, it is well established that many GIST cells do not die after the patient starts taking imatinib, but rather enter a quiescent state in which they no longer reproduce by cell division. If the patient stops taking the drug, these cells may re-enter the cell division cycle and cause resumed tumor growth.
In her new paper Dr. Duensing and colleagues traced the mechanisms inside the cell that allow the GIST cell to become quiescent and thereby avoid apoptosis. These dormant GIST cells can reactivate and start growing again if the patient stops taking imatinib. They might also develop resistance to imatinib while the patient is still taking it. Therefore, understanding the “quiescence response†is important to patient treatment outcomes.
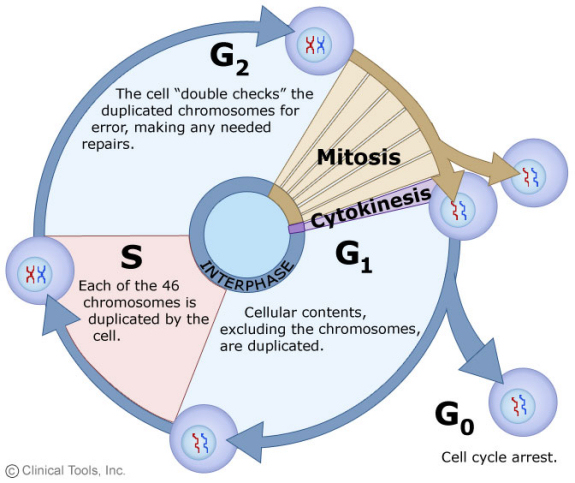
The cell cycle is the normal process by which cells divide. Before birth and during childhood, cell division allows the child to grow. In adults, cell division is needed only to heal wounds or to replace cells that have died. In tumors, however, abnormal cell division allows the cancer to grow. In the diagram below, the cell cycle is illustrated by the steps in the circle, while the normal resting state is shown as G0, or cell cycle arrest. The mitotic count, used in classifying GIST risk of recurrence, is a measure of the number of cells in the M phase, mitosis. Cells exit the cell cycle and remain in G0 to fulfill their functions in the body without any growth. This G0 state is quiescence.
In her research, Dr. Duensing used the GIST cell line GIST882 to study how imatinib may cause GIST cells to become quiescent. She found that cells enter quiescence when CDH1 moves into the cell nucleus, SKP2 is reduced by degradation, and p27Kip1 is increased. In untreated cells, activated KIT protein maintained high levels of SKP2. When Dr. Duensing artificially increased levels of SKP2, GIST882 cells resisted entering quiescence.
Dr. Duensing also examined GIST cells from 28 primary tumor samples from GIST patients to measure levels of SKP2. She determined that the samples from tumors classified as high risk to recur or metastasize (based on tumor size and mitotic rate) showed higher levels of SKP2. This implies that high-risk GIST cells would be less likely to become quiescent when the patient takes imatinib, due to the higher levels of SKP2. Manipulation of SKP2 might therefore be a way to enhance GIST response to imatinib.
Dr. Duensing and her lab team continue to investigate these factors in GIST cell response to treatment. Here are some of their goals:
- They want to know whether quiescence and apoptosis are mutually exclusive choices, or whether quiescent cells may still undergo imatinib-induced death.
- They want to determine whether other GIST cell lines show the same responses as GIST882, which bears an uncommon exon 13 mutation of the KIT gene.
- They want to find out how long GIST cells can remain quiescent without dying, and to unravel the contribution of the PI3K pathway in cell fate.
- Dr. Duensing and her colleagues hope to find a way to force quiescent GIST cells back into the cell cycle, where they may be more susceptible to apoptosis.
New Findings on Proteasome Inhibition To Combat GIST
The newest research results from Dr. Duensing’s lab were just presented at the 2008 Connective Tissue Oncology Society (CTOS) meeting in London on November 15. Dr. Duensing’s work was selected as an oral presentation there, indicating its importance. Her paper is abstract #35008 entitled “The Proteolytic Machinery As Novel Therapeutic Target In Gastrointestinal Stromal Tumors (GISTS).”
Building on the earlier work summarized above, Dr. Duensing stated in her presentation “The results suggest that untreated GIST cells keep H2AX at low levels in a pathway that involves oncogenic KIT and the ubiquitin-proteasome machinery. Based on these results we tested the proteasome inhibitor bortezomib (Velcade) for its antitumour activity in GIST. Bortezomib was able to induce apoptosis in a dose- and time-dependent manner. Effective concentrations were similar to those in use in multiple myeloma. Similar to imatinib-treated cells, histone H2AX was upregulated after bortezomib treatment. However, it appears that bortezomib has a dual-action mechanism in GIST, because it also dramatically downregulated KIT protein levels through transcriptional inhibition. Importantly, bortezomib also effectively induced apoptosis in short term cultures of an imatinib-resistant GIST. Taken together, these results suggest an active ubiquitin-proteosome system is crucial for GIST cell survival and proliferation and that it can be exploited to develop novel therapeutic approaches.â€
Bortezomib (brand name Velcade) is a proteasome inhibitor drug that is FDA-approved for treatment of multiple myeloma and mantle cell lymphoma. The proteasome can be thought of as the cell’s recycling center where proteins that have fulfilled their immediate function are broken down (degraded). Bortezomib’s mode of action is to prevent the proteasome from disassembling proteins, so the proteins stay active.
Therefore, Dr. Duensing’s research indicates that bortezomib allows H2AX to build up to higher levels, thereby allowing the GIST cell to die through apoptosis. In addition, it seems that bortezomib can also reduce the production of KIT protein on which the cells depend so much, revealing a dual mechanism of action. Her results using cell cultures are exciting, but the effect will need to be tested in vivo (in living animals) before it could be tested in GIST patients. However, the fact that bortezomib is an approved drug would suggest that testing might proceed more rapidly than for an unapproved drug.
Thanks to Dr. Duensing for her work on GIST! We eagerly follow her lab’s new findings and their treatment implications for GIST patients. You can donate to Dr. Duensing’s research though GSI’s Donate page.

